|
This order makes qualitative sense based on the kinds and extents of motion available to atoms and molecules in the three phases. K), whereas S° for water vapor is 188.8 J/(mol Online calculator, figures and tables with dynamic (absolute) and kinematic viscosity for air at temperatures ranging -100 to 1600C (-150 to 2900F) and at pressures ranging 1 to 10 000 bara (14.5 - 145000 psia) - SI and Imperial Units.For instance, S° for liquid water is 70.0 J/(mol Where n and m are the coefficients found in the balanced chemical equation of the reaction.\), for substances with approximately the same molar mass and number of atoms, S° values fall in the order S°(gas) > S°(liquid) > S°(solid). 
The entropy change of a reaction where the reactants and products are in their standard state can be determined using the following equation: (Source: UC Davis ChemWiki by University of California\CC-BY-SA-3.0) Standard Entropy Change of a Reaction, Δ S° At absolute zero there is only 1 microstate possible (1 1 ) and ln(1) 0. Temperature of a Single Substance.” This is a generalized plot of entropy versus temperature for a single substance. The standard entropy of a substance is its entropy at 1 atm pressure. These large increases occur due to sudden increased molecular mobility and larger available volumes associated with the phase changes.įigure 18.3 “Entropy vs. This can be seen in Figure 18.3 “Entropy vs. Temperature of a Single Substance.” Large jumps in entropy occur at the phase changes: solid to liquid and liquid to gas. Enthalpy of formation, Gibbs function of formation, and absolute entropy at. for an element in its standard state is zero. the standard state is the form in which the element exists (is more stable) under condition of 1 bar and at the temperature of interest (usually 25C). The standard molar entropy of any substance increases as the temperature increases. a pure substance in a condensed state (liquid or solid), the standard state is the pure liquid or solid under 1 bar pressure.Gases tend to have much larger standard molar enthalpies than liquids, and liquids tend to have larger values than solids, when comparing the same or similar substances.There are more possible arrangements of atoms in space for larger, more complex molecules, increasing the number of possible microstates. Thus the change in the internal energy of the system is related to the change in entropy, the absolute temperature, and the PV work done. Like energy and enthalpy, entropy is a state function. wrev PV, we can express Equation 13.4.3 as follows: U qrev + wrev TS PV. The absolute entropy of one mole of a substance in its standard state (P 1 atm and specified T) is called the standard molar entropy, S. Standard entropy is the absolute entropy of a substance at 1 atm and 25C. Larger, more complex molecules have higher standard molar enthalpy values than smaller or simpler molecules. Recognizing that the work done in a reversible process at constant pressure is.The idea that the entropy change for a pure substance goes to zero as. Several trends emerge from standard molar entropy data: This page titled 11.3: The Third Law is shared under a CC BY-SA 4.0 license and was authored, remixed, and/or curated by Paul Ellgen via source content that was edited to the style and standards of the LibreTexts platform a detailed edit history is available upon request. Table 18.1c Standard Molar Entropies of Selected Solids at 298 K Solid Table 18.1b Standard Molar Entropies of Selected Liquids at 298 K Liquid Table 18.1a Standard Molar Entropies of Selected Gases at 298 K Gas These values have been tabulated, and selected substances are listed in Table 18.1a to c “Standard Molar Entropies of Selected Substances at 298 K”. The standard molar entropy, S°, is the entropy of 1 mole of a substance in its standard state, at 1 atm of pressure. 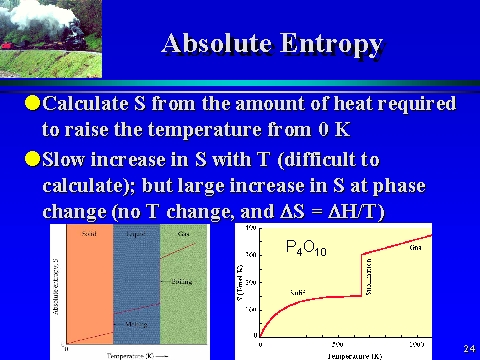 
Assume the change is reversible and the temperature remains constant. Determine the change in entropy (in J/K) of water when 425 kJ of heat is applied to it at 50☌. The solid line refers to the entropy of strontium in its normal standard state at 1 atm pressure.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
